A provirus is a virus genome that is integrated into the DNA of a host cell. In the case of bacterial viruses (bacteriophages), proviruses are often referred to as prophages. However, proviruses are distinctly different from prophages and these terms should not be used interchangeably. Unlike prophages, proviruses do not excise themselves from the host genome when the host cell is stressed.
Mouse mammary tumor virus (MMTV) is a milk-transmitted retrovirus like the HTL viruses, HI viruses, and BLV. It belongs to the genus Betaretrovirus. MMTV was formerly known as Bittner virus, and previously the "milk factor", referring to the extra-chromosomal vertical transmission of murine breast cancer by adoptive nursing, demonstrated in 1936, by John Joseph Bittner while working at the Jackson Laboratory in Bar Harbor, Maine. Bittner established the theory that a cancerous agent, or "milk factor", could be transmitted by cancerous mothers to young mice from a virus in their mother's milk. The majority of mammary tumors in mice are caused by mouse mammary tumor virus.
HHV Latency Associated Transcript is a length of RNA which accumulates in cells hosting long-term, or latent, Human Herpes Virus (HHV) infections. The LAT RNA is produced by genetic transcription from a certain region of the viral DNA. LAT regulates the viral genome and interferes with the normal activities of the infected host cell.

Herpesviridae is a large family of DNA viruses that cause infections and certain diseases in animals, including humans. The members of this family are also known as herpesviruses. The family name is derived from the Greek word ἕρπειν, referring to spreading cutaneous lesions, usually involving blisters, seen in flares of herpes simplex 1, herpes simplex 2 and herpes zoster (shingles). In 1971, the International Committee on the Taxonomy of Viruses (ICTV) established Herpesvirus as a genus with 23 viruses among four groups. As of 2020, 115 species are recognized, all but one of which are in one of the three subfamilies. Herpesviruses can cause both latent and lytic infections.
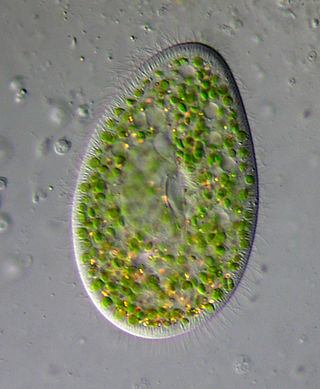
Paramecium bursaria is a species of ciliate found in marine and brackish waters. It has a mutualistic endosymbiotic relationship with green algae called Zoochlorella. About 700 Chlorella cells live inside the protist's cytoplasm and provide it with food, while the Paramecium provides the algae with movement and protection. P. bursaria is 80-150 μm long, with a wide oral groove, two contractile vacuoles, and a single micronucleus as well as a single macronucleus. P. bursaria is the only species of Paramecium that forms symbiotic relationships with algae, and it is often used in biology classrooms both as an example of a protozoan and also as an example of symbiosis.
The murine leukemia viruses are retroviruses named for their ability to cause cancer in murine (mouse) hosts. Some MLVs may infect other vertebrates. MLVs include both exogenous and endogenous viruses. Replicating MLVs have a positive sense, single-stranded RNA (ssRNA) genome that replicates through a DNA intermediate via the process of reverse transcription.

Herpes simplex virus1 and 2, also known by their taxonomic names Human alphaherpesvirus 1 and Human alphaherpesvirus 2, are two members of the human Herpesviridae family, a set of viruses that produce viral infections in the majority of humans. Both HSV-1 and HSV-2 are very common and contagious. They can be spread when an infected person begins shedding the virus.
Phycodnaviridae is a family of large (100–560 kb) double-stranded DNA viruses that infect marine or freshwater eukaryotic algae. Viruses within this family have a similar morphology, with an icosahedral capsid. As of 2014, there were 33 species in this family, divided among 6 genera. This family belongs to a super-group of large viruses known as nucleocytoplasmic large DNA viruses. Evidence was published in 2014 suggesting that specific strains of Phycodnaviridae might infect humans rather than just algal species, as was previously believed. Most genera under this family enter the host cell by cell receptor endocytosis and replicate in the nucleus. Phycodnaviridae play important ecological roles by regulating the growth and productivity of their algal hosts. Algal species such Heterosigma akashiwo and the genus Chrysochromulina can form dense blooms which can be damaging to fisheries, resulting in losses in the aquaculture industry. Heterosigma akashiwo virus (HaV) has been suggested for use as a microbial agent to prevent the recurrence of toxic red tides produced by this algal species. Phycodnaviridae cause death and lysis of freshwater and marine algal species, liberating organic carbon, nitrogen and phosphorus into the water, providing nutrients for the microbial loop.

A virus is a tiny infectious agent that reproduces inside the cells of living hosts. When infected, the host cell is forced to rapidly produce thousands of identical copies of the original virus. Unlike most living things, viruses do not have cells that divide; new viruses assemble in the infected host cell. But unlike simpler infectious agents like prions, they contain genes, which allow them to mutate and evolve. Over 4,800 species of viruses have been described in detail out of the millions in the environment. Their origin is unclear: some may have evolved from plasmids—pieces of DNA that can move between cells—while others may have evolved from bacteria.

A virus is a submicroscopic infectious agent that replicates only inside the living cells of an organism. Viruses infect all life forms, from animals and plants to microorganisms, including bacteria and archaea. Viruses are found in almost every ecosystem on Earth and are the most numerous type of biological entity. Since Dmitri Ivanovsky's 1892 article describing a non-bacterial pathogen infecting tobacco plants and the discovery of the tobacco mosaic virus by Martinus Beijerinck in 1898, more than 11,000 of the millions of virus species have been described in detail. The study of viruses is known as virology, a subspeciality of microbiology.

HBx is a hepatitis B viral protein. It is 154 amino acids long and interferes with transcription, signal transduction, cell cycle progress, protein degradation, apoptosis and chromosomal stability in the host. It forms a heterodimeric complex with its cellular target protein, and this interaction dysregulates centrosome dynamics and mitotic spindle formation. It interacts with DDB1 redirecting the ubiquitin ligase activity of the CUL4-DDB1 E3 complexes, which are intimately involved in the intracellular regulation of DNA replication and repair, transcription and signal transduction.

Nudiviruses are a family of animal viruses that constitute the family Nudiviridae. Insects and marine crustaceans serve as natural hosts. There are 11 species in this family, assigned to 4 genera. Diseases associated with this family include: death in larvae, chronic disease in adults.
Mamavirus is a large and complex virus in the Group I family Mimiviridae. The virus is exceptionally large, and larger than many bacteria. Mamavirus and other mimiviridae belong to nucleocytoplasmic large DNA virus (NCLDVs) family. Mamavirus can be compared to the similar complex virus mimivirus; mamavirus was so named because it is similar to but larger than mimivirus.

Cafeteria roenbergensis virus (CroV) is a giant virus that infects the marine bicosoecid flagellate Cafeteria roenbergensis, a member of the microzooplankton community.
A giant virus, sometimes referred to as a girus, is a very large virus, some of which are larger than typical bacteria. All known giant viruses belong to the phylum Nucleocytoviricota.
Acanthocystis turfacea chlorella virus 1 (ATCV-1), also called Chlorovirus ATCV-1 or Chlorella virus ATCV-1 is a species of giant double-stranded DNA virus in the genus Chlorovirus.
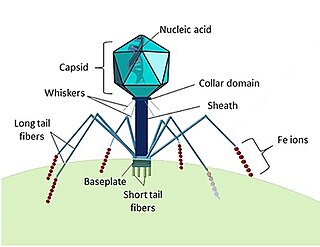
Marine viruses are defined by their habitat as viruses that are found in marine environments, that is, in the saltwater of seas or oceans or the brackish water of coastal estuaries. Viruses are small infectious agents that can only replicate inside the living cells of a host organism, because they need the replication machinery of the host to do so. They can infect all types of life forms, from animals and plants to microorganisms, including bacteria and archaea.

Varidnaviria is a realm of viruses that includes all DNA viruses that encode major capsid proteins that contain a vertical jelly roll fold. The major capsid proteins (MCP) form into pseudohexameric subunits of the viral capsid, which stores the viral deoxyribonucleic acid (DNA), and are perpendicular, or vertical, to the surface of the capsid. Apart from this, viruses in the realm also share many other characteristics, such as minor capsid proteins (mCP) with the vertical jelly roll fold, an ATPase that packages viral DNA into the capsid, and a DNA polymerase that replicates the viral genome.
Algal viruses are the viruses infecting photosynthetic single-celled eukaryotes, algae. As of 2020, there were 61 viruses known to infect algae. Algae are integral components of aquatic food webs and drive nutrient cycling, so the viruses infecting algal populations also impacts the organisms and nutrient cycling systems that depend on them. Thus, these viruses can have significant, worldwide economic and ecological effects. Their genomes varied between 4.4 to 560 kilobase pairs (kbp) long and used double-stranded Deoxyribonucleic Acid (dsDNA), double-stranded Ribonucleic Acid (dsRNA), single-stranded Deoxyribonucleic Acid (ssDNA), and single-stranded Ribonucleic Acid (ssRNA). The viruses ranged between 20 and 210 nm in diameter. Since the discovery of the first algae-infecting virus in 1979, several different techniques have been used to find new viruses infecting algae and it seems that there are many algae-infecting viruses left to be discovered
Virivore comes from the English prefix viro- meaning virus, derived from the Latin word for poison, and the suffix -vore from the Latin word vorare, meaning to eat, or to devour; therefore, a virivore is an organism that consumes viruses. Virivory is a well-described process in which organisms, primarily heterotrophic protists, but also some metazoans consume viruses.












