A DNA virus is a virus that has a genome made of deoxyribonucleic acid (DNA) that is replicated by a DNA polymerase. They can be divided between those that have two strands of DNA in their genome, called double-stranded DNA (dsDNA) viruses, and those that have one strand of DNA in their genome, called single-stranded DNA (ssDNA) viruses. dsDNA viruses primarily belong to two realms: Duplodnaviria and Varidnaviria, and ssDNA viruses are almost exclusively assigned to the realm Monodnaviria, which also includes some dsDNA viruses. Additionally, many DNA viruses are unassigned to higher taxa. Reverse transcribing viruses, which have a DNA genome that is replicated through an RNA intermediate by a reverse transcriptase, are classified into the kingdom Pararnavirae in the realm Riboviria.
Virus classification is the process of naming viruses and placing them into a taxonomic system similar to the classification systems used for cellular organisms.
Baltimore classification is a system used to classify viruses based on their manner of messenger RNA (mRNA) synthesis. By organizing viruses based on their manner of mRNA production, it is possible to study viruses that behave similarly as a distinct group. Seven Baltimore groups are described that take into consideration whether the viral genome is made of deoxyribonucleic acid (DNA) or ribonucleic acid (RNA), whether the genome is single- or double-stranded, and whether the sense of a single-stranded RNA genome is positive or negative.

Virophages are small, double-stranded DNA viral phages that require the co-infection of another virus. The co-infecting viruses are typically giant viruses. Virophages rely on the viral replication factory of the co-infecting giant virus for their own replication. One of the characteristics of virophages is that they have a parasitic relationship with the co-infecting virus. Their dependence upon the giant virus for replication often results in the deactivation of the giant viruses. The virophage may improve the recovery and survival of the host organism.

Mimivirus-dependent virus Sputnik is a subviral agent that reproduces in amoeba cells that are already infected by a certain helper virus; Sputnik uses the helper virus's machinery for reproduction and inhibits replication of the helper virus. It is known as a virophage, in analogy to the term bacteriophage.

Bidensovirus is a genus of single stranded DNA viruses that infect invertebrates. The species in this genus were originally classified in the family Parvoviridae but were moved to a new genus because of significant differences in the genomes.
A transpoviron is a plasmid-like genetic element found in the genomes of giant DNA viruses.
Tristromaviridae is a family of viruses. Archaea of the genera Thermoproteus and Pyrobaculum serve as natural hosts. Tristromaviridae is the sole family in the order Primavirales. There are two genera and three species in the family.

Mimivirus-dependent virus Zamilon, or Zamilon, is a virophage, a group of small DNA viruses that infect protists and require a helper virus to replicate; they are a type of satellite virus. Discovered in 2013 in Tunisia, infecting Acanthamoeba polyphaga amoebae, Zamilon most closely resembles Sputnik, the first virophage to be discovered. The name is Arabic for "the neighbour". Its spherical particle is 50–60 nm in diameter, and contains a circular double-stranded DNA genome of around 17 kb, which is predicted to encode 20 polypeptides. A related strain, Zamilon 2, has been identified in North America.
Polintons are large DNA transposons which contain genes with homology to viral proteins and which are often found in eukaryotic genomes. They were first discovered in the mid-2000s and are the largest and most complex known DNA transposons. Polintons encode up to 10 individual proteins and derive their name from two key proteins, a DNA polymerase and a retroviral-like integrase.

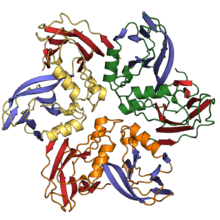
The jelly roll or Swiss roll fold is a protein fold or supersecondary structure composed of eight beta strands arranged in two four-stranded sheets. The name of the structure was introduced by Jane S. Richardson in 1981, reflecting its resemblance to the jelly or Swiss roll cake. The fold is an elaboration on the Greek key motif and is sometimes considered a form of beta barrel. It is very common in viral proteins, particularly viral capsid proteins. Taken together, the jelly roll and Greek key structures comprise around 30% of the all-beta proteins annotated in the Structural Classification of Proteins (SCOP) database.
Riboviria is a realm of viruses that includes all viruses that use a homologous RNA-dependent polymerase for replication. It includes RNA viruses that encode an RNA-dependent RNA polymerase, as well as reverse-transcribing viruses that encode an RNA-dependent DNA polymerase. RNA-dependent RNA polymerase (RdRp), also called RNA replicase, produces RNA from RNA. RNA-dependent DNA polymerase (RdDp), also called reverse transcriptase (RT), produces DNA from RNA. These enzymes are essential for replicating the viral genome and transcribing viral genes into messenger RNA (mRNA) for translation of viral proteins.
In virology, realm is the highest taxonomic rank established for viruses by the International Committee on Taxonomy of Viruses (ICTV), which oversees virus taxonomy. Six virus realms are recognized and united by specific highly conserved traits:

Duplodnaviria is a realm of viruses that includes all double-stranded DNA viruses that encode the HK97 fold major capsid protein. The HK97 fold major capsid protein is the primary component of the viral capsid, which stores the viral deoxyribonucleic acid (DNA). Viruses in the realm also share a number of other characteristics, such as an icosahedral capsid, an opening in the viral capsid called a portal, a protease enzyme that empties the inside of the capsid prior to DNA packaging, and a terminase enzyme that packages viral DNA into the capsid.

Monodnaviria is a realm of viruses that includes all single-stranded DNA viruses that encode an endonuclease of the HUH superfamily that initiates rolling circle replication of the circular viral genome. Viruses descended from such viruses are also included in the realm, including certain linear single-stranded DNA (ssDNA) viruses and circular double-stranded DNA (dsDNA) viruses. These atypical members typically replicate through means other than rolling circle replication.

Cressdnaviricota is a phylum of viruses with small, circular single-stranded DNA genomes and encoding rolling circle replication-initiation proteins with the N-terminal HUH endonuclease and C-terminal superfamily 3 helicase domains. While the replication-associated proteins are homologous among viruses within the phylum, the capsid proteins are very diverse and have presumably been acquired from RNA viruses on multiple independent occasions. Nevertheless, all cressdnaviruses for which structural information is available appear to contain the jelly-roll fold.

Bamfordvirae is a kingdom of viruses. This kingdom is recognized for its use of double jelly roll major capsid proteins. It was formerly known as the PRD1-adenovirus lineage. The kingdom is named after Dennis H. Bamford who first promoted the evolutionary unity of all viruses encoding double jelly-roll major capsid proteins.

An archaeal virus is a virus that infects and replicates in archaea, a domain of unicellular, prokaryotic organisms. Archaeal viruses, like their hosts, are found worldwide, including in extreme environments inhospitable to most life such as acidic hot springs, highly saline bodies of water, and at the bottom of the ocean. They have been also found in the human body. The first known archaeal virus was described in 1974 and since then, a large diversity of archaeal viruses have been discovered, many possessing unique characteristics not found in other viruses. Little is known about their biological processes, such as how they replicate, but they are believed to have many independent origins, some of which likely predate the last archaeal common ancestor (LACA).

Portogloboviridae is a family of dsDNA viruses that infect archaea. It is a proposed family of the realm Varidnaviria, but ICTV officially puts it as incertae sedis virus. Viruses in the family are related to Helvetiavirae. The capsid proteins of these viruses and their characteristics are of evolutionary importance for the origin of the other Varidnaviria viruses since they seem to retain primordial characters.

Adnaviria is a realm of viruses that includes archaeal viruses that have a filamentous virion and a linear, double-stranded DNA genome. The genome exists in A-form (A-DNA) and encodes a dimeric major capsid protein (MCP) that contains the SIRV2 fold, a type of alpha-helix bundle containing four helices. The virion consists of the genome encased in capsid proteins to form a helical nucleoprotein complex. For some viruses, this helix is surrounded by a lipid membrane called an envelope. Some contain an additional protein layer between the nucleoprotein helix and the envelope. Complete virions are long and thin and may be flexible or a stiff like a rod.