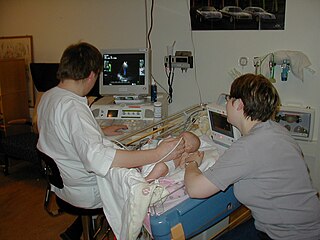
Medical ultrasound includes diagnostic techniques using ultrasound, as well as therapeutic applications of ultrasound. In diagnosis, it is used to create an image of internal body structures such as tendons, muscles, joints, blood vessels, and internal organs, to measure some characteristics or to generate an informative audible sound. The usage of ultrasound to produce visual images for medicine is called medical ultrasonography or simply sonography, or echography. The practice of examining pregnant women using ultrasound is called obstetric ultrasonography, and was an early development of clinical ultrasonography. The machine used is called an ultrasound machine, a sonograph or an echograph. The visual image formed using this technique is called an ultrasonogram, a sonogram or an echogram.
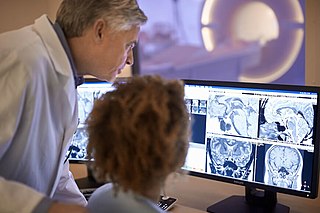
Radiology is the medical specialty that uses medical imaging to diagnose diseases and guide their treatment, within the bodies of humans and other animals. It began with radiography, but today it includes all imaging modalities, including those that use no ionizing electromagnetic radiation, as well as others that do, such as computed tomography (CT), fluoroscopy, and nuclear medicine including positron emission tomography (PET). Interventional radiology is the performance of usually minimally invasive medical procedures with the guidance of imaging technologies such as those mentioned above.

Mammography is the process of using low-energy X-rays to examine the human breast for diagnosis and screening. The goal of mammography is the early detection of breast cancer, typically through detection of characteristic masses or microcalcifications.
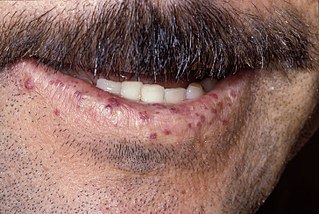
Hereditary hemorrhagic telangiectasia (HHT), also known as Osler–Weber–Rendu disease and Osler–Weber–Rendu syndrome, is a rare autosomal dominant genetic disorder that leads to abnormal blood vessel formation in the skin, mucous membranes, and often in organs such as the lungs, liver, and brain.

Electrical impedance tomography (EIT) is a noninvasive type of medical imaging in which the electrical conductivity, permittivity, and impedance of a part of the body is inferred from surface electrode measurements and used to form a tomographic image of that part. Electrical conductivity varies considerably among various biological tissues or the movement of fluids and gases within tissues. The majority of EIT systems apply small alternating currents at a single frequency, however, some EIT systems use multiple frequencies to better differentiate between normal and suspected abnormal tissue within the same organ.
Computed tomography laser mammography (CTLM) is the trademark of Imaging Diagnostic Systems, Inc. for its optical tomographic technique for female breast imaging.

Dermatoscopy also known as dermoscopy or epiluminescence microscopy, is the examination of skin lesions with a dermatoscope. It is a tool similar to a camera to allow for inspection of skin lesions unobstructed by skin surface reflections. The dermatoscope consists of a magnifier, a light source, a transparent plate and sometimes a liquid medium between the instrument and the skin. The dermatoscope is often handheld, although there are stationary cameras allowing the capture of whole body images in a single shot. When the images or video clips are digitally captured or processed, the instrument can be referred to as a digital epiluminescence dermatoscope. The image is then analyzed automatically and given a score indicating how dangerous it is. This technique is useful to dermatologists and skin cancer practitioners in distinguishing benign from malignant (cancerous) lesions, especially in the diagnosis of melanoma.

Breast cancer screening is the medical screening of asymptomatic, apparently healthy women for breast cancer in an attempt to achieve an earlier diagnosis. The assumption is that early detection will improve outcomes. A number of screening tests have been employed, including clinical and self breast exams, mammography, genetic screening, ultrasound, and magnetic resonance imaging.

Tomosynthesis, also digital tomosynthesis (DTS), is a method for performing high-resolution limited-angle tomography at radiation dose levels comparable with projectional radiography. It has been studied for a variety of clinical applications, including vascular imaging, dental imaging, orthopedic imaging, mammographic imaging, musculoskeletal imaging, and chest imaging.
Daniel B. Kopans, MD, FACR is a radiologist specializing in mammography and other forms of breast imaging.
Electromagnetic navigation bronchoscopy (ENB) is a medical procedure utilizing electromagnetic technology designed to localize and guide endoscopic tools or catheters through the bronchial pathways of the lung. Using a virtual, three-dimensional (3D) bronchial map from a recently computed tomography (CT) chest scan and disposable catheter set, physicians are able to navigate to a desired location within the lung to biopsy lesions, stage lymph nodes, insert markers to guide radiotherapy or guide brachytherapy catheters.
Computer-aided simple triage (CAST) are computerized methods or systems that assist physicians in initial interpretation and classification of medical images. CAST is a sub-class of computer-aided diagnosis (CAD). CAST software systems perform a fully automatic initial triage (classification) of diagnostic medical imaging studies. CAST is primarily intended for emergency diagnostic imaging, where a prompt diagnosis of critical, life-threatening condition is required.
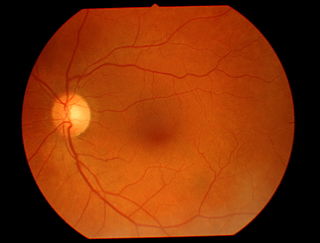
Teleophthalmology is a branch of telemedicine that delivers eye care through digital medical equipment and telecommunications technology. Today, applications of teleophthalmology encompass access to eye specialists for patients in remote areas, ophthalmic disease screening, diagnosis and monitoring; as well as distant learning.
Dynamic angiothermography (DATG) is a technique for the diagnosis of breast cancer. This technique, though springing from the previous conception of thermography, is based on a completely different principle. DATG records the temperature variations linked to the vascular changes in the breast due to angiogenesis. The presence, change, and growth of tumors and lesions in breast tissue change the vascular network in the breast. Consequently, through measuring the vascular structure over time, DATG effectively monitors the change in breast tissue due to tumors and lesions. It is currently used in combination with other techniques for diagnosis of breast cancer. This diagnostic method is a low-cost one compared with other techniques.
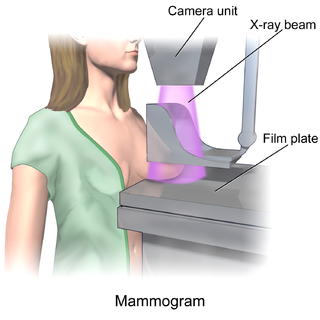
In medicine, breast imaging is a sub-speciality of diagnostic radiology that involves imaging of the breasts for screening or diagnostic purposes. There are various methods of breast imaging using a variety of technologies as described in detail below. Traditional screening and diagnostic mammography uses x-ray technology and has been the mainstay of breast imaging for many decades. Breast tomosynthesis is a relatively new digital x-ray mammography technique that produces multiple image slices of the breast similar to, but distinct from, computed tomography (CT). Xeromammography and galactography are somewhat outdated technologies that also use x-ray technology and are now used infrequently in the detection of breast cancer. Breast ultrasound is another technology employed in diagnosis and screening that can help differentiate between fluid filled and solid lesions, an important factor to determine if a lesion may be cancerous. Breast MRI is a technology typically reserved for high-risk patients and patients recently diagnosed with breast cancer. Lastly, scintimammography is used in a subgroup of patients who have abnormal mammograms or whose screening is not reliable on the basis of using traditional mammography or ultrasound.
In the field of medicine, radiomics is a method that extracts a large number of features from medical images using data-characterisation algorithms. These features, termed radiomic features, have the potential to uncover tumoral patterns and characteristics that fail to be appreciated by the naked eye. The hypothesis of radiomics is that the distinctive imaging features between disease forms may be useful for predicting prognosis and therapeutic response for various cancer types, thus providing valuable information for personalized therapy. Radiomics emerged from the medical fields of radiology and oncology and is the most advanced in applications within these fields. However, the technique can be applied to any medical study where a pathological process can be imaged.
Fiona Jane Gilbert is a Scottish radiologist and academic.
Ronald Marc Summers is an American radiologist and senior investigator at the Diagnostic Radiology Department at the NIH Clinical Center in Bethesda, Maryland. He is chief of the Clinical Image Processing Service and directs the Imaging Biomarkers and Computer-Aided Diagnosis (CAD) Laboratory. A researcher in the field of radiology and computer-aided diagnosis, he has co-authored over 500 journal articles and conference proceedings papers and is a coinventor on 12 patents. His lab has conducted research applying artificial intelligence and deep learning to radiology.

Aidoc Medical is an Israeli technology company that develops computer-aided simple triage and notification systems. Aidoc has obtained FDA and CE mark approval for its stroke, pulmonary embolism, cervical fracture, intracranial hemorrhage, intra-abdominal free gas, and incidental pulmonary embolism algorithms.
Medical open network for AI (MONAI) is an open-source, community-supported framework for Deep learning (DL) in healthcare imaging. MONAI provides a collection of domain-optimized implementations of various DL algorithms and utilities specifically designed for medical imaging tasks. MONAI is used in research and industry, aiding the development of various medical imaging applications, including image segmentation, image classification, image registration, and image generation.