
Fusobacterium is a genus of obligate anaerobic, Gram-negative, non-sporeforming bacteria belonging to Gracilicutes. Individual cells are slender, rod-shaped bacilli with pointed ends. Fusobacterium was discovered in 1900 by Courmont and Cade and is common in the flora of humans.

Agrobacterium is a genus of Gram-negative bacteria established by H. J. Conn that uses horizontal gene transfer to cause tumors in plants. Agrobacterium tumefaciens is the most commonly studied species in this genus. Agrobacterium is well known for its ability to transfer DNA between itself and plants, and for this reason it has become an important tool for genetic engineering.
Mycobacterium avium subspecies paratuberculosis (MAP) is an obligate pathogenic bacterium in the genus Mycobacterium. It is often abbreviated M. paratuberculosis or M. avium ssp. paratuberculosis. It is the causative agent of Johne's disease, which affects ruminants such as cattle, and suspected causative agent in human Crohn's disease and rheumatoid arthritis. The type strain is ATCC 19698.
Staphylococcus hominis is a coagulase-negative member of the bacterial genus Staphylococcus, consisting of Gram-positive, spherical cells in clusters. It occurs very commonly as a harmless commensal on human and animal skin and is known for producing thioalcohol compounds that contribute to body odour. Like many other coagulase-negative staphylococci, S. hominis may occasionally cause infection in patients whose immune systems are compromised, for example by chemotherapy or predisposing illness.

Lacticaseibacillus casei is an organism that belongs to the largest genus in the family Lactobacillaceae, a lactic acid bacteria (LAB), that was previously classified as Lactobacillus casei. This bacteria has been identified as facultatively anaerobic or microaerophilic, acid-tolerant, non-spore-forming bacteria.

Lacticaseibacillus rhamnosus is a bacterium that originally was considered to be a subspecies of L. casei, but genetic research found it to be a separate species in the L. casei clade, which also includes L. paracasei and L. zeae. It is a short Gram-positive homofermentative facultative anaerobic non-spore-forming rod that often appears in chains. Some strains of L. rhamnosus bacteria are being used as probiotics, and are particularly useful in treating infections of the female urogenital tract, most particularly very difficult to treat cases of bacterial vaginosis. The species Lacticaseibacillus rhamnosus and Limosilactobacillus reuteri are commonly found in the healthy female genito-urinary tract and are helpful to regain control of dysbiotic bacterial overgrowth during an active infection. L. rhamnosus sometimes is used in dairy products such as fermented milk and as non-starter-lactic acid bacterium (NSLAB) in long-ripened cheese. While frequently considered a beneficial organism, L. rhamnosus may not be as beneficial to certain subsets of the population; in rare circumstances, especially those primarily involving weakened immune system or infants, it may cause endocarditis. Despite the rare infections caused by L. rhamnosus, the species is included in the list of bacterial species with qualified presumed safety (QPS) status of the European Food Safety Agency.

Mycobacteroides abscessus is a species of rapidly growing, multidrug-resistant, nontuberculous mycobacteria (NTM) that is a common soil and water contaminant. Although M. abscessus most commonly causes chronic lung infection and skin and soft tissue infection (SSTI), it can also cause infection in almost all human organs, mostly in patients with suppressed immune systems. Amongst NTM species responsible for disease, infection caused by M. abscessus complex are more difficult to treat due to antimicrobial drug resistance.
Fusobacterium nucleatum is a Gram-negative, anaerobic oral bacterium, commensal to the human oral cavity, that plays a role in periodontal disease. This organism is commonly recovered from different monocultured microbial and mixed infections in humans and animals. In health and disease, it is a key component of periodontal plaque due to its abundance and its ability to coaggregate with other bacteria species in the oral cavity.
Porphyromonas gingivalis belongs to the phylum Bacteroidota and is a nonmotile, Gram-negative, rod-shaped, anaerobic, pathogenic bacterium. It forms black colonies on blood agar.

Cronobacter sakazakii, which before 2007 was named Enterobacter sakazakii, is an opportunistic Gram-negative, rod-shaped, pathogenic bacterium that can live in very dry places, otherwise known as xerotolerance. C. sakazakii utilizes a number of genes to survive desiccation and this xerotolerance may be strain specific. The majority of C. sakazakii cases are adults but low-birth-weight preterm neonatal and older infants are at the highest risk. The pathogen is a rare cause of invasive infection in infants, with historically high case fatality rates (40–80%).
Treponema denticola is a Gram-negative, obligate anaerobic, motile and highly proteolytic spirochete bacterium. It is one of four species of oral spirochetes to be reliably cultured, the others being Treponema pectinovorum, Treponema socranskii and Treponema vincentii. T. denticola dwells in a complex and diverse microbial community within the oral cavity and is highly specialized to survive in this environment. T. denticola is associated with the incidence and severity of human periodontal disease. Treponema denticola is one of three bacteria that form the Red Complex, the other two being Porphyromonas gingivalis and Tannerella forsythia. Together they form the major virulent pathogens that cause chronic periodontitis. Having elevated T. denticola levels in the mouth is considered one of the main etiological agents of periodontitis. T. denticola is related to the syphilis-causing obligate human pathogen, Treponema pallidum subsp. pallidum. It has also been isolated from women with bacterial vaginosis.

Streptococcus zooepidemicus is a Lancefield group C streptococcus that was first isolated in 1934 by P. R. Edwards, and named Animal pyogens A. It is a mucosal commensal and opportunistic pathogen that infects several animals and humans, but most commonly isolated from the uterus of mares. It is a subspecies of Streptococcus equi, a contagious upper respiratory tract infection of horses, and shares greater than 98% DNA homology, as well as many of the same virulence factors.

Mycoplasma mycoides is a bacterial species of the genus Mycoplasma in the class Mollicutes. This microorganism is a parasite that lives in ruminants. Mycoplasma mycoides comprises two subspecies, mycoides and capri, which infect cattle and small ruminants such as goats respectively.
Lautropia mirabilis is a Gram-negative, facultatively anaerobic, oxidase- and catalase-positive, motile bacterium of the genus Lautropia and family Burkholderiaceae, isolated from the mouth of children who were infected with human immunodeficiency virus.
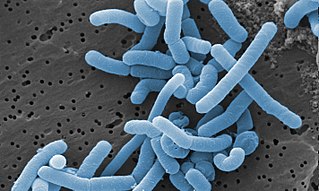
Lacticaseibacillus paracasei (commonly abbreviated as Lc. paracasei) is a gram-positive, homofermentative species of lactic acid bacteria that are commonly used in dairy product fermentation and as probiotic cultures. Lc. paracasei is a bacterium that operates by commensalism. It is commonly found in many human habitats such as human intestinal tracts and mouths as well as sewages, silages, and previously mentioned dairy products. The name includes morphology, a rod-shaped bacterium with a width of 2.0 to 4.0μm and length of 0.8 to 1.0μm.
Campylobacter showae is a Gram-negative, chemoheterotrophic, microaerophilic, motile bacteria belonging to the Campylobacter Genus. The type strain of this species, SU A4, was first isolated from plaque samples taken from the gingival crevices of the human oral cavity but has since also been found in colonic tissues and stool. Since its discovery, C. showae has been implicated in various medical conditions including Crohn's disease, periodontitis, inflammatory bowel disease, and ulcerative colitis due to its pathogenic nature.
Cryptobacterium curtum is a Gram-positive anaerobic rod bacteria isolated from human mouths.
Treponema socranskii was isolated from gum swabs of people with periodontitis and clinically-induced periodontitis. It is a motile, helically coiled, obligate anaerobe that grows best at 37 °C, and is a novel member of its genus because of its ability to ferment molecules that other Treponema species cannot. T. socranskii’s growth is positively correlated with gingival inflammation, which indicates that it is a leading cause of gingivitis and periodontitis.
Actinobacillus equuli is a gram-negative, non-motile rod bacteria from the family Pasteurellaceae.
Actinomyces massiliensis is an anaerobic, mesophilic, Gram-positive bacterium originally isolated from a human blood sample and belonging to the genus Actinomyces.








