
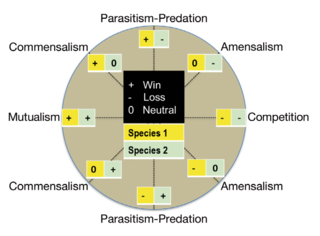
An endosymbiont or endobiont is any organism that lives within the body or cells of another organism most often, though not always, in a mutualistic relationship. (The term endosymbiosis is from the Greek: ἔνδον endon "within", σύν syn "together" and βίωσις biosis "living".) Examples are nitrogen-fixing bacteria, which live in the root nodules of legumes, single-cell algae inside reef-building corals and bacterial endosymbionts that provide essential nutrients to insects.

Corals are colonial marine invertebrates within the class Anthozoa of the phylum Cnidaria. They typically form compact colonies of many identical individual polyps. Coral species include the important reef builders that inhabit tropical oceans and secrete calcium carbonate to form a hard skeleton.
The dinoflagellates are a monophyletic group of single-celled eukaryotes constituting the phylum Dinoflagellata and are usually considered protists. Dinoflagellates are mostly marine plankton, but they also are common in freshwater habitats. Their populations vary with sea surface temperature, salinity, and depth. Many dinoflagellates are photosynthetic, but a large fraction of these are in fact mixotrophic, combining photosynthesis with ingestion of prey.

Coral bleaching is the process when corals become white due to various stressors, such as changes in temperature, light, or nutrients. Bleaching occurs when coral polyps expel the zooxanthellae that live inside their tissue, causing the coral to turn white. The zooxanthellae are photosynthetic, and as the water temperature rises, they begin to produce reactive oxygen species. This is toxic to the coral, so the coral expels the zooxanthellae. Since the zooxanthellae produce the majority of coral colouration, the coral tissue becomes transparent, revealing the coral skeleton made of calcium carbonate. Most bleached corals appear bright white, but some are blue, yellow, or pink due to pigment proteins in the coral.
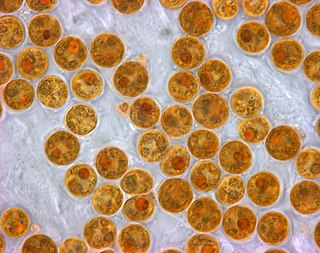
Zooxanthellae is a colloquial term for single-celled dinoflagellates that are able to live in symbiosis with diverse marine invertebrates including demosponges, corals, jellyfish, and nudibranchs. Most known zooxanthellae are in the genus Symbiodinium, but some are known from the genus Amphidinium, and other taxa, as yet unidentified, may have similar endosymbiont affinities. The true Zooxanthella K.brandt is a mutualist of the radiolarian Collozoum inerme and systematically placed in Peridiniales. Another group of unicellular eukaryotes that partake in similar endosymbiotic relationships in both marine and freshwater habitats are green algae zoochlorellae.
Symbiotic bacteria are bacteria living in symbiosis with another organism or each other. For example, rhizobia living in root nodules of legumes provide nitrogen fixing activity for these plants.

Aiptasia is a genus of a symbiotic cnidarian belonging to the class Anthozoa. Aiptasia is a widely distributed genus of temperate and tropical sea anemones of benthic lifestyle typically found living on mangrove roots and hard substrates. These anemones, as well as many other cnidarian species, often contain symbiotic dinoflagellate unicellular algae of the genus Symbiodinium living inside nutritive cells. The symbionts provide food mainly in the form of lipids and sugars produced from photosynthesis to the host while the hosts provides inorganic nutrients and a constant and protective environment to the algae. Species of Aiptasia are relatively weedy anemones able to withstand a relatively wide range of salinities and other water quality conditions. In the case of A. pallida and A. pulchella, their hardiness coupled with their ability to reproduce very quickly and out-compete other species in culture gives these anemones the status of pest from the perspective of coral reef aquarium hobbyists. These very characteristics make them easy to grow in the laboratory and thus they are extensively used as model organisms for scientific study. In this respect, Aiptasia have contributed a significant amount of knowledge regarding cnidarian biology, especially human understanding of cnidarian-algal symbioses, a biological phenomenon crucial to the survival of corals and coral reef ecosystems. The dependence of coral reefs on the health of the symbiosis is dramatically illustrated by the devastating effects experienced by corals due to the loss of algal symbionts in response to environmental stress, a phenomenon known as coral bleaching.
Cyanobionts are cyanobacteria that live in symbiosis with a wide range of organisms such as terrestrial or aquatic plants; as well as, algal and fungal species. They can reside within extracellular or intracellular structures of the host. In order for a cyanobacterium to successfully form a symbiotic relationship, it must be able to exchange signals with the host, overcome defense mounted by the host, be capable of hormogonia formation, chemotaxis, heterocyst formation, as well as possess adequate resilience to reside in host tissue which may present extreme conditions, such as low oxygen levels, and/or acidic mucilage. The most well-known plant-associated cyanobionts belong to the genus Nostoc. With the ability to differentiate into several cell types that have various functions, members of the genus Nostoc have the morphological plasticity, flexibility and adaptability to adjust to a wide range of environmental conditions, contributing to its high capacity to form symbiotic relationships with other organisms. Several cyanobionts involved with fungi and marine organisms also belong to the genera Richelia, Calothrix, Synechocystis, Aphanocapsa and Anabaena, as well as the species Oscillatoria spongeliae. Although there are many documented symbioses between cyanobacteria and marine organisms, little is known about the nature of many of these symbioses. The possibility of discovering more novel symbiotic relationships is apparent from preliminary microscopic observations.
Chromera velia, also known as a "chromerid", is a unicellular photosynthetic organism in the superphylum Alveolata. It is of interest in the study of apicomplexan parasites, specifically their evolution and accordingly, their unique vulnerabilities to drugs.
The resilience of coral reefs is the biological ability of coral reefs to recover from natural and anthropogenic disturbances such as storms and bleaching episodes. Resilience refers to the ability of biological or social systems to overcome pressures and stresses by maintaining key functions through resisting or adapting to change. Reef resistance measures how well coral reefs tolerate changes in ocean chemistry, sea level, and sea surface temperature. Reef resistance and resilience are important factors in coral reef recovery from the effects of ocean acidification. Natural reef resilience can be used as a recovery model for coral reefs and an opportunity for management in marine protected areas (MPAs).
The hologenome theory of evolution recasts the individual animal or plant as a community or a "holobiont" – the host plus all of its symbiotic microbes. Consequently, the collective genomes of the holobiont form a "hologenome". Holobionts and hologenomes are structural entities that replace misnomers in the context of host-microbiota symbioses such as superorganism, organ, and metagenome. Variation in the hologenome may encode phenotypic plasticity of the holobiont and can be subject to evolutionary changes caused by selection and drift, if portions of the hologenome are transmitted between generations with reasonable fidelity. One of the important outcomes of recasting the individual as a holobiont subject to evolutionary forces is that genetic variation in the hologenome can be brought about by changes in the host genome and also by changes in the microbiome, including new acquisitions of microbes, horizontal gene transfers, and changes in microbial abundance within hosts. Although there is a rich literature on binary host–microbe symbioses, the hologenome concept distinguishes itself by including the vast symbiotic complexity inherent in many multicellular hosts. For recent literature on holobionts and hologenomes published in an open access platform, see the following reference.

Pocillopora damicornis, commonly known as the cauliflower coral or lace coral, is a species of stony coral in the family Pocilloporidae. It is native to tropical and subtropical parts of the Indian and Pacific Oceans.

Orbicella faveolata, commonly known as mountainous star coral, is a colonial stony coral in the family Merulinidae. Orbicella faveolata is native to the coral coast of the Caribbean Sea and the Gulf of Mexico and is listed as "endangered" by the International Union for Conservation of Nature. O. faveolata was formerly known as Montastraea faveolata.
Durusdinium trenchii is an endosymbiotic dinoflagellate, a unicellular alga which commonly resides in the tissues of tropical corals. It has a greater tolerance to fluctuations in water temperatures than do other species in the genus. It was named for the marine biologist R. K. Trench.
Microbial symbiosis in marine animals was not discovered until 1981. In the time following, symbiotic relationships between marine invertebrates and chemoautotrophic bacteria have been found in a variety of ecosystems, ranging from shallow coastal waters to deep-sea hydrothermal vents. Symbiosis is a way for marine organisms to find creative ways to survive in a very dynamic environment. They are different in relation to how dependent the organisms are on each other or how they are associated. It is also considered a selective force behind evolution in some scientific aspects. The symbiotic relationships of organisms has the ability to change behavior, morphology and metabolic pathways. With increased recognition and research, new terminology also arises, such as holobiont, which the relationship between a host and its symbionts as one grouping. Many scientists will look at the hologenome, which is the combined genetic information of the host and its symbionts. These terms are more commonly used to describe microbial symbionts.

All animals on Earth form associations with microorganisms, including protists, bacteria, archaea, fungi, and viruses. In the ocean, animal–microbial relationships were historically explored in single host–symbiont systems. However, new explorations into the diversity of marine microorganisms associating with diverse marine animal hosts is moving the field into studies that address interactions between the animal host and a more multi-member microbiome. The potential for microbiomes to influence the health, physiology, behavior, and ecology of marine animals could alter current understandings of how marine animals adapt to change, and especially the growing climate-related and anthropogenic-induced changes already impacting the ocean environment.
Robert Kent Trench was an American Biologist who was a professor at the University of California, Santa Barbara. His research considered corals and symbiotic algae, with a focus on the adaption of zooxanthellae. He was awarded the 1994 International Society of Endocytobiology Miescher-Ishida Prize.

Mary Alice Coffroth is an American marine biologist who is a professor at the State University of New York at Buffalo. She is known for her use of molecular tools to examined coral larval ecology, recruitment and cnidarian-dinoflagellate symbiosis.

Symbiodiniaceae is a family of marine dinoflagellates notable for their symbiotic associations with reef-building corals, sea anemones, jellyfish, marine sponges, giant clams, acoel flatworms, and other marine invertebrates. Symbiotic Symbiodiniaceae are sometimes colloquially referred to as Zooxanthellae, though the latter term can be interpreted to include other families of symbiotic algae as well. While many Symbiodiniaceae species are endosymbionts, others are free living in the water column or sediment.
Durusdinium is a genus of dinoflagellate algae within the family Symbiodiniaceae. Durusdinium can be free living, or can form symbiotic associations with hard corals. Members of the genus have been documented in reef-building corals of the Indian and Pacific oceans, as well as the Caribbean. Prior to 2018, Durusdinium were classified as Symbiodinium Clade D.


























