Basidiomycota is one of two large divisions that, together with the Ascomycota, constitute the subkingdom Dikarya within the kingdom Fungi. Members are known as basidiomycetes. More specifically, Basidiomycota includes these groups: agarics, puffballs, stinkhorns, bracket fungi, other polypores, jelly fungi, boletes, chanterelles, earth stars, smuts, bunts, rusts, mirror yeasts, and Cryptococcus, the human pathogenic yeast.

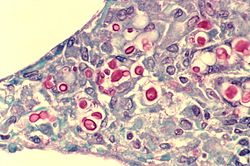
Cryptococcus is a genus of fungi in the family Cryptococcaceae that includes both yeasts and filamentous species. The filamentous, sexual forms or teleomorphs were formerly classified in the genus Filobasidiella, while Cryptococcus was reserved for the yeasts. Most yeast species formerly referred to Cryptococcus have now been placed in different genera. The name Cryptococcus comes from the Greek for "hidden sphere". Some Cryptococcus species cause a disease called cryptococcosis.

Amphotericin B is an antifungal medication used for serious fungal infections and leishmaniasis. The fungal infections it is used to treat include mucormycosis, aspergillosis, blastomycosis, candidiasis, coccidioidomycosis, and cryptococcosis. For certain infections it is given with flucytosine. It is typically given intravenously.

Cryptococcosis is a potentially fatal fungal infection of mainly the lungs, presenting as a pneumonia, and brain, where it appears as a meningitis. Cough, difficulty breathing, chest pain and fever are seen when the lungs are infected. When the brain is infected, symptoms include headache, fever, neck pain, nausea and vomiting, light sensitivity and confusion or changes in behavior. It can also affect other parts of the body including skin, where it may appear as several fluid-filled nodules with dead tissue.

Fluconazole is an antifungal medication used for a number of fungal infections. This includes candidiasis, blastomycosis, coccidioidomycosis, cryptococcosis, histoplasmosis, dermatophytosis, and tinea versicolor. It is also used to prevent candidiasis in those who are at high risk such as following organ transplantation, low birth weight babies, and those with low blood neutrophil counts. It is given either by mouth or by injection into a vein.

The bacterial capsule is a large structure common to many bacteria. It is a polysaccharide layer that lies outside the cell envelope, and is thus deemed part of the outer envelope of a bacterial cell. It is a well-organized layer, not easily washed off, and it can be the cause of various diseases.
Intracellular parasites are microparasites that are capable of growing and reproducing inside the cells of a host. They are also called intracellular pathogens.
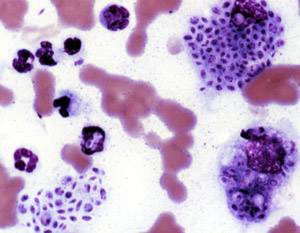
Sporotrichosis, also known as rose handler's disease, is a fungal infection that may be localised to skin, lungs, bone and joint, or become systemic. It presents with firm painless nodules that later ulcerate. Following initial exposure to Sporothrix schenckii, the disease typically progresses over a period of a week to several months. Serious complications may develop in people who have a weakened immune system.

Flucytosine, also known as 5-fluorocytosine (5-FC), is an antifungal medication. It is specifically used, together with amphotericin B, for serious Candida infections and cryptococcosis. It may be used by itself or with other antifungals for chromomycosis. Flucytosine is used by mouth and by injection into a vein.
Immune reconstitution inflammatory syndrome (IRIS) is a condition seen in some cases of HIV/AIDS or immunosuppression, in which the immune system begins to recover, but then responds to a previously acquired opportunistic infection with an overwhelming inflammatory response that paradoxically makes the symptoms of infection worse.
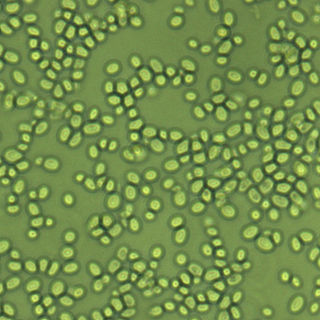
Nakaseomyces glabratus is a species of haploid yeast of the genus Nakaseomyces, previously known as Candida glabrata. Despite the fact that no sexual life cycle has been documented for this species, N. glabratus strains of both mating types are commonly found. N. glabrata is generally a commensal of human mucosal tissues, but in today's era of wider human immunodeficiency from various causes, N. glabratus is often the second or third most common cause of candidiasis as an opportunistic pathogen. Infections caused by N. glabratus can affect the urogenital tract or even cause systemic infections by entrance of the fungal cells in the bloodstream (Candidemia), especially prevalent in immunocompromised patients.

In biology, a phagolysosome, or endolysosome, is a cytoplasmic body formed by the fusion of a phagosome with a lysosome in a process that occurs during phagocytosis. Formation of phagolysosomes is essential for the intracellular destruction of microorganisms and pathogens. It takes place when the phagosome's and lysosome's membranes 'collide', at which point the lysosomal contents—including hydrolytic enzymes—are discharged into the phagosome in an explosive manner and digest the particles that the phagosome had ingested. Some products of the digestion are useful materials and are moved into the cytoplasm; others are exported by exocytosis.

Cryptococcus gattii, formerly known as Cryptococcus neoformans var. gattii, is an encapsulated yeast found primarily in tropical and subtropical climates. Its teleomorph is Filobasidiella bacillispora, a filamentous fungus belonging to the class Tremellomycetes.
Pathogenic fungi are fungi that cause disease in humans or other organisms. Although fungi are eukaryotic, many pathogenic fungi are microorganisms. Approximately 300 fungi are known to be pathogenic to humans; their study is called "medical mycology". Fungal infections are estimated to kill more people than either tuberculosis or malaria—about two million people per year.

OSU-03012 (AR-12) is a celecoxib derivative with anticancer and anti-microbial activity. Unlike celecoxib, OSU-03012 does not inhibit COX, but inhibits several other important enzymes instead which may be useful in the treatment of some forms of cancer, When combined with PDE5 inhibitors such as sildenafil or tadalafil, OSU-03012 was found to show enhanced anti-tumour effects in cell culture.
Vomocytosis is the cellular process by phagocytes expel live organisms that they have engulfed without destroying the organism. Vomocytosis is one of many methods used by cells to expel internal materials into their external environment, yet it is distinct in that both the engulfed organism and host cell remain undamaged by expulsion. As engulfed organisms are released without being destroyed, vomocytosis has been hypothesized to be utilized by pathogens as an escape mechanism from the immune system. The exact mechanisms, as well as the repertoire of cells that utilize this mechanism, are currently unknown, yet interest in this unique cellular process is driving continued research with the hopes of elucidating these unknowns.

Arturo Casadevall is a Bloomberg Distinguished Professor of Molecular Microbiology & Immunology and Infectious Diseases at the Johns Hopkins Bloomberg School of Public Health and Johns Hopkins School of Medicine, and the Alfred and Jill Sommer Professor and Chair of the W. Harry Feinstone Department of Molecular Microbiology and Immunology at the Johns Hopkins Bloomberg School of Public Health. He is an internationally recognized expert in infectious disease research, with a focus on fungal and bacterial pathogenesis and basic immunology of antibody structure-function. He was elected a member of the National Academy of Sciences in 2022.

Joseph Heitman is an American physician-scientist focused on research in genetics, microbiology, and infectious diseases. He is the James B. Duke Professor and Chair of the Department of Molecular Genetics and Microbiology at Duke University School of Medicine.
Naganishia albida is a species of fungus in the family Filobasidiaceae. It is currently only known from its yeast state. The species was originally isolated from the air in Japan, and has subsequently been isolated from dry moss in Portugal, grasshoppers in Portugal, and tubercular lungs.
Chronic meningitis is a long-lasting inflammation of the membranes lining the brain and spinal cord. By definition, the duration of signs, symptoms and inflammation in chronic meningitis last longer than 4 weeks. Infectious causes are a leading cause and the infectious organisms responsible for chronic meningitis are different than the organisms that cause acute infectious meningitis. Tuberculosis and the fungi cryptococcus are leading causes worldwide. Chronic meningitis due to infectious causes are more common in those who are immunosuppressed, including those with HIV infection or in children who are malnourished. Chronic meningitis sometimes has a more indolent course than acute meningitis with symptoms developing more insidiously and slowly. Also, some of the infectious agents that cause chronic infectious meningitis such as mycobacterium tuberculosis, many fungal species and viruses are difficult to isolate from the cerebrospinal fluid making diagnosis challenging. No cause is identified during initial evaluation in one third of cases. Magnetic resonance imaging (MRI) of the brain is more sensitive than computed tomography and may show radiological signs that suggest chronic meningitis, however no radiological signs are considered pathognomonic or characteristic. MRI is also normal in many cases further limiting its diagnostic utility.